cdna lab - PRS BioSciences Confocal GFP Probes
Insect general odor binding proteins (GOBPs) have long been thought to bind and transport plant host volatiles to olfactory receptors on the dendritic membrane of olfactory neurons. Recent studies indicate that they can also bind to female sex pheromones. In the present study, two GOBP genes, AipsGOBP1 and AipsGOBP2, were cloned from the adult antennae of Agrotis ipsilon. Tissue expression profiles indicated that both are antennae specific and more abundant in female than male antennae. Temporal expression profiles showed that both AipsGOBP1 and AipsGOBP2 began to express in antennae 3 days before adult pupal emergence, and reached their highest expression level 3 and 4 days after adult emergence, respectively. Mating increased its expression on female antennae but reduced its expression on male antennae.
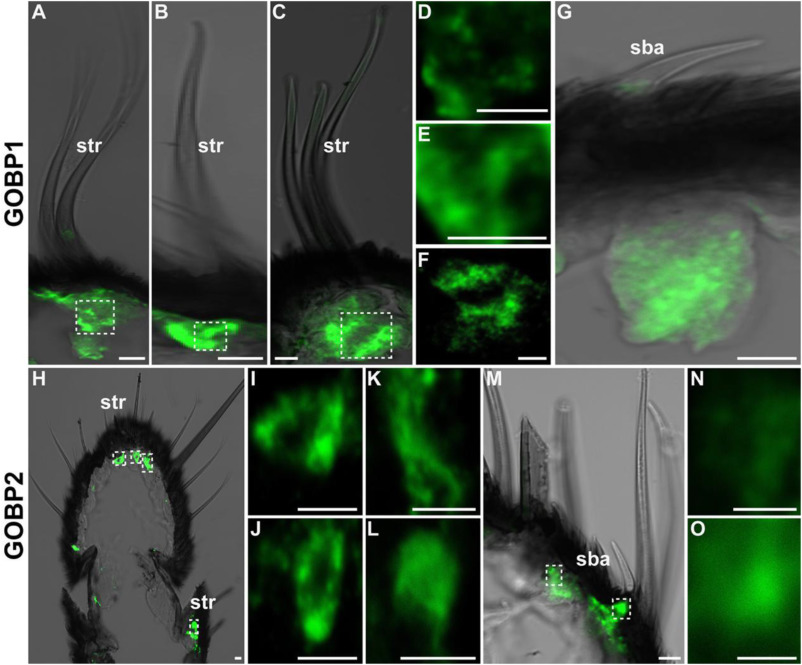
Hybridization and in situ immunolocation demonstrated that both AipsGOBP1 and AipsGOBP2 are expressed and co-localized in sensilla basiconica and sensilla trichodea of both sexes. AipsGOBP2 exhibited a high in vitro binding affinity with the two main components of sex pheromones Z7-12: Ac and Z9-14: Ac and the four plant volatiles cis-3-hexen-1-ol, oleic acid, dibutyl phthalate and β-caryophyllene. with Ki values lower than 5 μM. AipsGOBP1, on the other hand, showed average binding affinities with the five A. ipsilon sex pheromones and six plant volatiles. AipsGOBP2 also showed a broader ligand binding spectrum and higher ligand binding affinity than AipsGOBP1 with the aldehyde and alcohol sex pheromones of Lepidoptera species tested. Taken together, our results indicate that AipsGOBP2 may play a more important role than AipsGOBP1 in binding sex and volatile pheromones from the host plant.
Insect antennae, as the most important olfactory organ, play a crucial role in locating mates, detecting food sources, and suitable oviposition sites (Visser, 1986; Field et al., 2000). There are several types of sensillas located on insect antennae, which can express specific olfactory proteins to detect chemical or semiochemical signals such as pheromones and plant volatiles from the outside world (Schneider, 1964; Sachse and Krieger, 2011; Leal, 2013). In general, pheromones and plant volatiles diffuse into the sensilla through individual pores that penetrate the cuticular surface (Steinbrecht, 1997). However, the hydrophobic characteristic of pheromones and plant volatiles makes these odors practically insoluble in the environment of the lymph sensillum to reach the olfactory receptors (OR) (Pelosi and Maida, 1995). Therefore, these hydrophobic chemicals are believed to be dissolved and transported by soluble carrier proteins, which are called odor-binding proteins (OBPs) (Pelosi and Maida, 1995; Kaissling, 2004; Zhou, 2010). After activating the ORs, the odor molecules are subsequently degraded by odor degrading enzymes (ODE) (Vogt and Riddiford, 1981; Leal, 2013). Insect OBPs are generally synthesized and secreted by non-neuronal helper cells (trichogenic and tormogenic cells) in the antennal sensilla. They exist in extremely high concentration (up to 10 mM) in lymph sensillum, which implies their important physiological roles in both chemical communication and insect survival (Vogt and Riddiford, 1981; Klein, 1987; Pelosi et al. , 2018).
Pheromone-binding proteins (PBPs) and general odor-binding proteins (GOBPs) are two major subfamilies of OBPs in Lepidoptera species (Zhou, 2010). The first PBP insect was identified in 1981 from the giant silk moth Antheraea polyphemus (Vogt and Riddiford, 1981); Ten years later, GOBP insects were identified for the first time in six species of Lepidoptera (Vogt et al., 1991). So far, at least 82 GOBPs and 91 PBPs from 52 Lepidoptera species are registered in the NCBI GenBank database.
Insect PBPs are believed to be involved in the binding of hydrophobic sex pheromones to membrane-associated pheromone receptors (PR) for the initiation of specific behaviors (Ziegelberger, 1996; Leal, 2005; Zhou, 2010; Stengl, 2010 ). In situ hybridization and immunocytochemical localization analyzes showed that PBPs are predominantly synthesized by pheromone-sensitive trichoid sensilla support cells (Steinbrecht et al., 1992; Zhang et al., 2001; Forstner et al., 2006; Gu et al., 2013). The experimental evidence for Lepidopteran PBPs in the binding of sex pheromones is verified both by in vitro binding studies (Zhou et al., 2009; Gu et al., 2013; Liu et al., 2013; Sun et al., 2013a ), in live genomic editing CRISPR-Cas9 studies (Zhu et al., 2016a; Dong et al., 2017b; Ye et al., 2017) and RNAi study (Dong et al., 2017a). Furthermore, some PBPs were shown to interact with PRs to improve the sensitivity and specificity of PRs to sex pheromones (Van den Berg and Ziegelberger, 1991; Groβe-Wilde et al., 2007; Forstner et al., 2009; Sun et al., 2013b; Chang et al., 2015).
Compared to PBPs, GOBPs are more abundant in female antennae and are located in the odor-sensitive sensilla basiconica in general (Laue et al., 1994; Steinbrecht et al., 1995; Laue, 2000). Functional studies of GOBP moths revealed their putative roles in interaction with plant semiochemicals. For example, GOBP2 from Manduca sexta (Feng and Prestwich, 1997), GOBP1-2 from Spodoptera exigua (Liu et al., 2015a), GOBP2 from Spodoptera litura (Liu et al., 2015b), GOBP2 from Loxostege sticticalis (Yin et al., 2012) and GOBP2 from Chilo suppressalis (Gong et al., 2009) can bind strongly with the volatiles of the host plant. However, growing evidence showed that insect GOBPs are also abundantly expressed in sex pheromone-sensitive sensilla trichodea and are capable of binding to insect sex pheromones with high affinities. For example, GOBP2 from Mamestra brassicae (Jacquin-Joly et al., 2000), GOBP2 from Bombyx mori (He et al., 2010), GOBP2 from M. sexta (Feng and Prestwich, 1997; Nardi et al., 2003) , GOBP1-2 from C. suppressalis (Khuhro et al., 2017), GOBP2 from S. exigua (Liu et al., 2015a) and GOBP1-2 from S. litura (Liu et al., 2015b) may bind strongly to the sex pheromones, suggesting their supposed roles in sex pheromone recognition. More interestingly, a recent study of Plutella xylostella GOBPs indicated that they may be involved in attracting larvae to their pheromones to search for better at