Indicaid Antigen 25 tests box
What is this rapid test kit?
This rapid test for sale is available to qualified healthcare professionals as well as business owners seeking a stock of rapid tests for on-site testing. This rapid test kit provides an easy-to-read result for an active infection in approximately 10 minutes. This rapid test kit was also recently approved for shallow nasal sampling, making sampling much more efficient and comfortable.
Revelation:
This test is not FDA-approved or approved; the test is FDA-approved under an Emergency Use Authorization (EUA) for use by labs certified under the CLIA that meet the requirements to perform tests of moderate, high, or exempt complexity. This test is authorized for use at the Point of Care (POC), i.e. in patient care facilities operating under a CLIA Certificate of Distance, Certificate of Compliance, or Certificate of Accreditation.

Accuracy Summary:
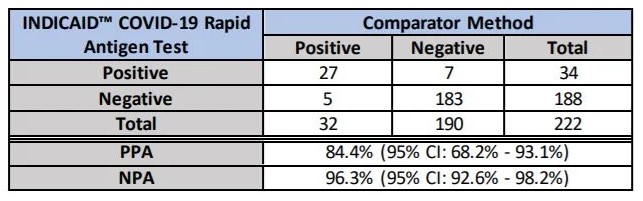
Positive Percent Agreement: 93.1%
Negative Percent Agreement: 98.2%
CPT CODE: 87426
What's in this kit?
This Rapid Antigen Nasal Test Kit is sold in boxes of 25 individual test cassettes and contains the following:
(25) Rapid Test Cassettes
- Foil bag testing device with one test strip that: comes in a plastic device case.
(25) Extraction Vials/Caps
- The extraction vial contains 400 l extraction buffer solution.
(25) Front Nasal Swabs
- Swabs for collecting shallow nasal samples
(1) User manual insert
Clinical Accuracy Findings
Intended Use of the Rapid Antigen Nasal Test Kit
- This antigen test is a lateral flow immunochromatographic test intended for the qualitative detection of the nucleocapsid protein antigen of SARS-CoV-2 in nasopharyngeal swab specimens collected directly or collected in BD universal transport media, of individuals suspected of having by their healthcare provider within five days of symptom get started.
- This test is authorized for use at the Point of Care (POC), i.e. in patient care facilities operating under a CLIA certificate of waiver, certificate of compliance, or Certificate of Accreditation The results are for the identification of the SARS-CoV-2 nucleocapsid protein antigen. the antigen is generally detectable in anterior nasal swabs during the acute phase of infection.
- Positive results indicate the presence of viral antigens, but the clinical correlation with the patient history and other diagnostic information is needed to determine infection status. Positive results do not rule out bacterial infection or co-infection with other viruses. The agent has detected: may not be the definitive cause of the disease. United States Laboratories and Their Territories are required to report all results to the appropriate public health authorities.
- Negative results are suspected and will be confirmed with a molecular test, if necessary, for patient care can be performed. Negative results do not exclude SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions. Negative results should be considered in the context of a recent exposures, patient history and presence of clinical signs and symptoms consistent with .
- The Indicaid Antigen is intended for use by medical professionals or trained operators skilled in performing tests and trained clinical laboratory personnel or persons trained in a healthcare institution. The Indicaid antigen is for use only under the Emergency Use Authorization of the Food and Drug Administration.
Collection and Use Instructions
Procedural Notes
- Process the test sample immediately after collection.
- Use only the provided or recommended anterior nasal swabs for sampling.
- Collect the sample with safety gloves to avoid contamination.
- Do not touch the tip (specimen collection area) of the swab.
- Collect samples as soon as possible after the onset of symptoms.